Abstract
Background: Adolescent and young adult (AYA) patients (pts) with acute lymphoblastic leukemia (ALL) or lymphoblastic lymphoma (LBL) have good outcomes when treated on pediatric regimens but still face worse survival compared to younger children (Siegel JAMA Onc 2018). Pediatric patients who do not complete ALL treatment, particularly planned asparaginase (ASP), have inferior oncologic outcomes (Silverman Blood 1997). In the CALBG 10403 trial of a pediatric-inspired regimen in AYAs 17-39 years (yrs), only 39% of pts completed planned treatment (Stock Blood 2019). We aimed to describe the prevalence of treatment failure, reasons for treatment non-completion, and their effect on survival among patients treated on Dana-Farber Cancer Institute (DFCI) ALL Consortium protocols.
Methods: We identified Philadelphia chromosome negative (Ph-) pts aged 1-49 yrs treated on 4 sequential multi-center DFCI Consortium protocols between 2000 and 2018 (00-001, 05-001, 01-175, 06-254). Additional consecutive Ph- pts treated as per these protocols were identified by chart review. All pts were intended to receive 30 continuous weeks (wks) of ASP during consolidation. Treatment failure was defined as induction failure, induction death, relapse/progression on therapy, and remission death on therapy. Treatment non-completion was defined as transplant in first complete remission (CR), withdrawal due to toxicity or other reasons, loss to follow up, and ineligible status. Treatment completion was defined as no exit from treatment protocol before its completion, approximately 2 yrs from complete remission (CR). Per protocol, no patients were recommended for allogeneic hematopoietic stem cell transplant (HSCT) in CR1. ASP completion was defined as receiving at least 26 wks of ASP during consolidation (Silverman, Blood 2001). The protocols allowed for truncation of ASP for toxicity without exit from protocol. Median follow-up time was calculated using the reverse Kaplan Meier indicator. The chi square test was used to compare proportions between groups. Overall survival (OS) among groups was compared using the log rank test.
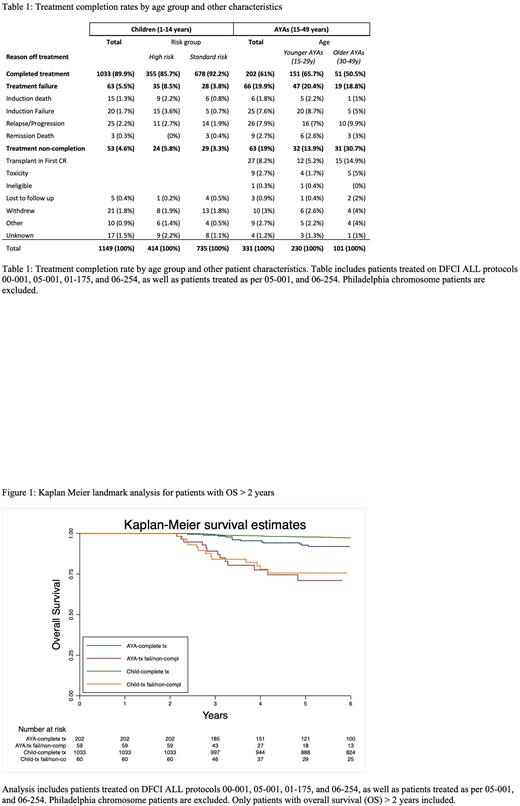
Results: A total of 1,480 pts were identified: 1,149 children (< 15 yrs, median age 4.5 yrs) and 331 AYAs (15-49 yrs, median age 24 yrs). Median follow-up was 7.9 yrs. Treatment completion rate was lower among AYAs than children (61.3% vs. 89.9%, p < 0.001) (Table 1). This was driven by higher treatment failure rates among AYAs than children (19.9% vs. 5.5%) as well as higher treatment non-completion rates (19.0% vs. 4.6%), primarily driven by transplants in CR1 among AYAs. Treatment completion rates were lower among older (30-49 yrs) AYAs compared to younger (15-29 yrs) AYAs (50.5% vs. 65.7%, p = 0.009). Among 1235 pts who completed treatment, 1086 (87.9%) received ³26 weeks of ASP. ASP completion rate was lower among AYAs vs. children (79.2% vs. 89.6%, p < 0.001). A higher percentage of AYAs discontinued ASP because of toxicity than children (11.9% vs. 5.5%, p = 0.01). In a landmark analysis of patients who survived and had follow-up for at least 2 years from diagnosis, AYAs who completed treatment (with or without ASP completion) had an estimated 5-year OS of 92.7% (95% Confidence Interval (CI): 87.5%, 95.8%), while children who completed treatment had a 5-year OS of 97.9% (95% CI: 96.7%, 98.6%) (Log-rank test p = 0.002) (Figure 1). Patients alive at 2 years who failed or did not complete treatment had worse survival outcomes with a 5-year OS of 70.5% (95% CI: 53.9%, 82.1%) among AYAs and 76.1% (95% CI: 62.2%, 85.5%) among children (Log-rank test p = 0.598)
Conclusions: AYAs in our cohort completed protocol treatment at higher rates than previously reported trials. Among AYAs who completed treatment protocol, oncologic outcomes were excellent (5-year OS above 90%) and comparable to (although statistically lower than) those of children. AYAs had higher treatment failure rates, likely due to more aggressive disease biology. AYAs had lower ASP completion rates and higher ASP discontinuation rates due to toxicity compared to children; the etiology and impact of that on survival will need to be further investigated. Introduction of novel agents to improve remission induction rates and prevent early relapse in AYAs might help further improve oncologic outcomes for this population.
Disclosures
Place:Servier: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; Novartis: Consultancy, Other: Meal, Research Funding. Silverman:Jazz Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees. Brunner:Taiho: Consultancy; Novartis: Consultancy, Research Funding; AstraZeneca: Research Funding; Keros Therapeutics: Consultancy; Janssen: Research Funding; GSK: Research Funding; Celgene/BMS: Consultancy, Research Funding; Acceleron: Honoraria; Agios: Honoraria; Takeda: Consultancy, Research Funding; Aprea: Research Funding. Stone:BerGenBio: Consultancy; Boston Pharmaceuticals: Consultancy; Kura Oncology: Consultancy; BMS: Consultancy; Jazz: Consultancy; Gemoab: Consultancy; Janssen: Consultancy; Novartis: Consultancy; OncoNova: Consultancy; GSK: Consultancy; Innate: Consultancy; Foghorn Therapeutics: Consultancy; Apteva: Consultancy; Aprea: Consultancy; Arog: Consultancy, Research Funding; Astellas: Consultancy; Elevate Bio: Consultancy; Epizyme: Consultancy; Syndax: Consultancy; Syntrix: Consultancy; Syros: Consultancy; Takeda: Consultancy; Actinium: Consultancy; Abbvie: Consultancy, Research Funding. DeAngelo:Amgen: Consultancy; Agios: Consultancy; Autolus: Consultancy; Forty-Seven: Consultancy; Incyte: Consultancy; Blueprint Medicines Corporation: Consultancy; Abbvie: Research Funding; Glycomimetics: Research Funding; Shire: Consultancy; Takeda: Consultancy; Novartis: Consultancy, Research Funding; Pfizer: Consultancy; Jazz Pharmaceuticals: Consultancy. Luskin:Pfizer: Honoraria; Abbvie: Research Funding; Novartis: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal